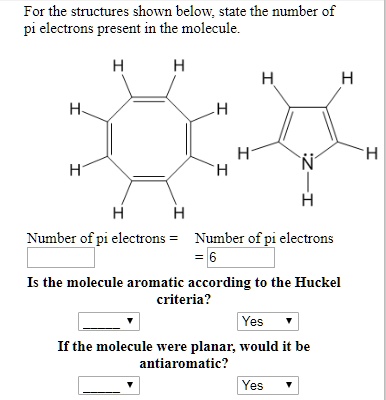
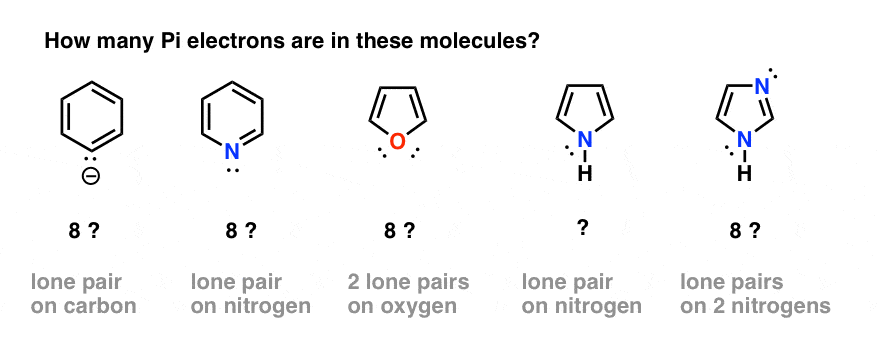
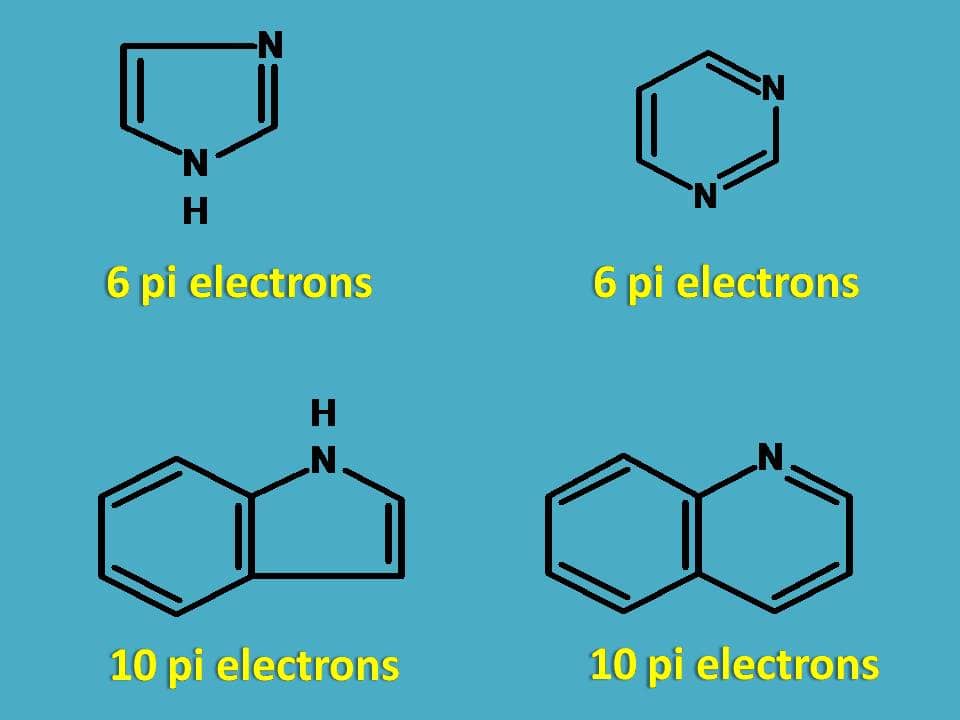
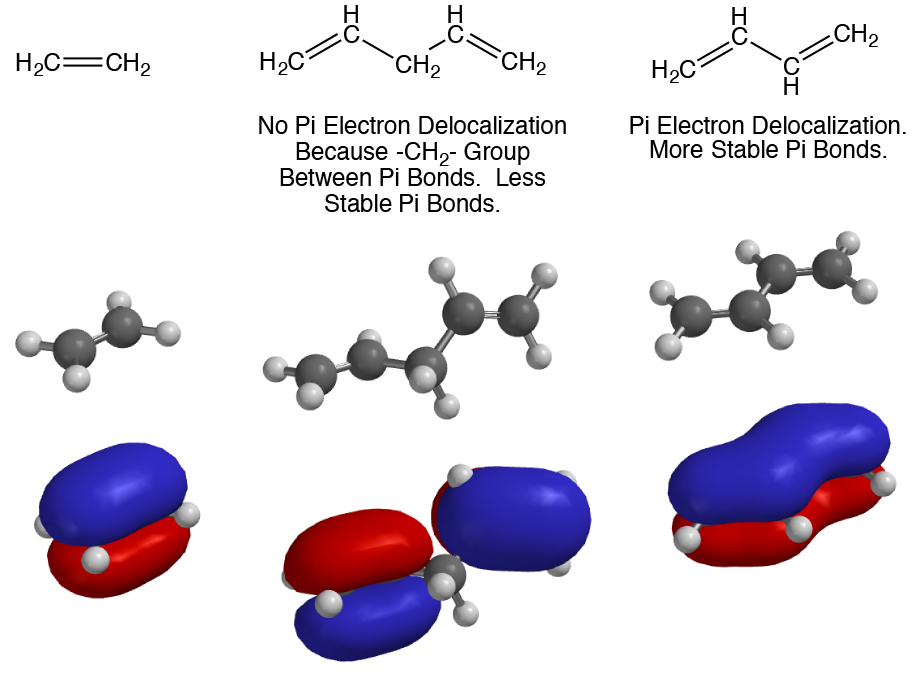
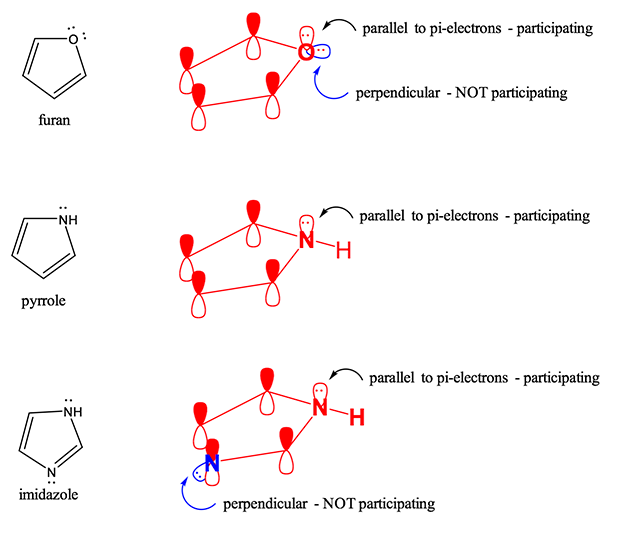
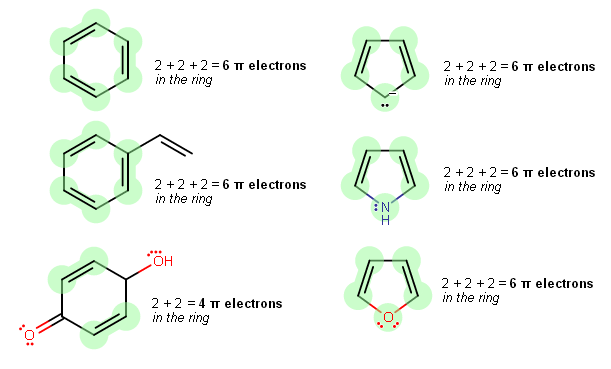
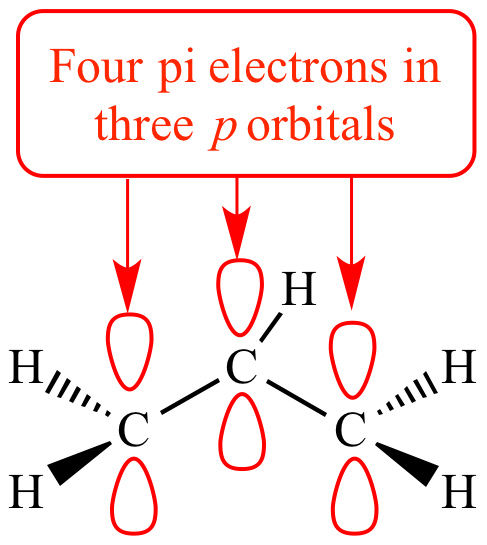
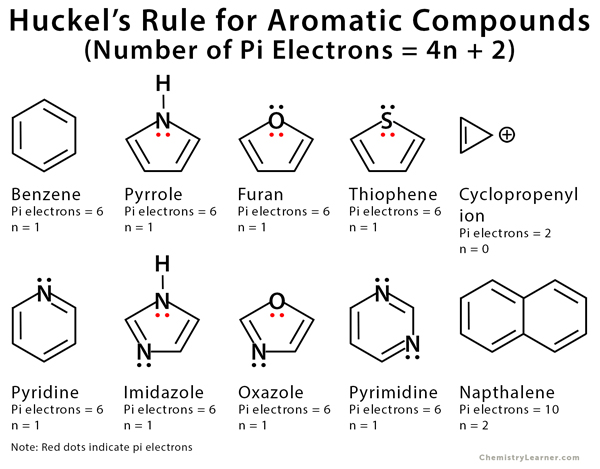
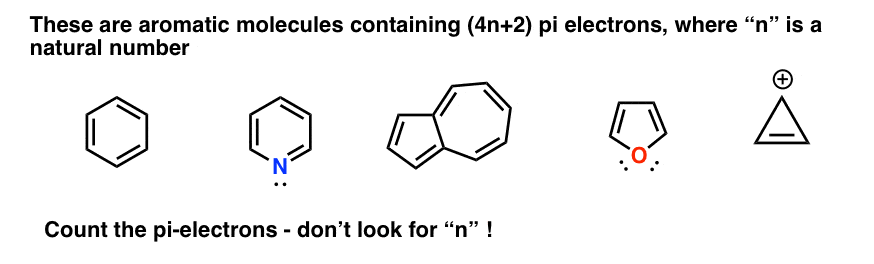
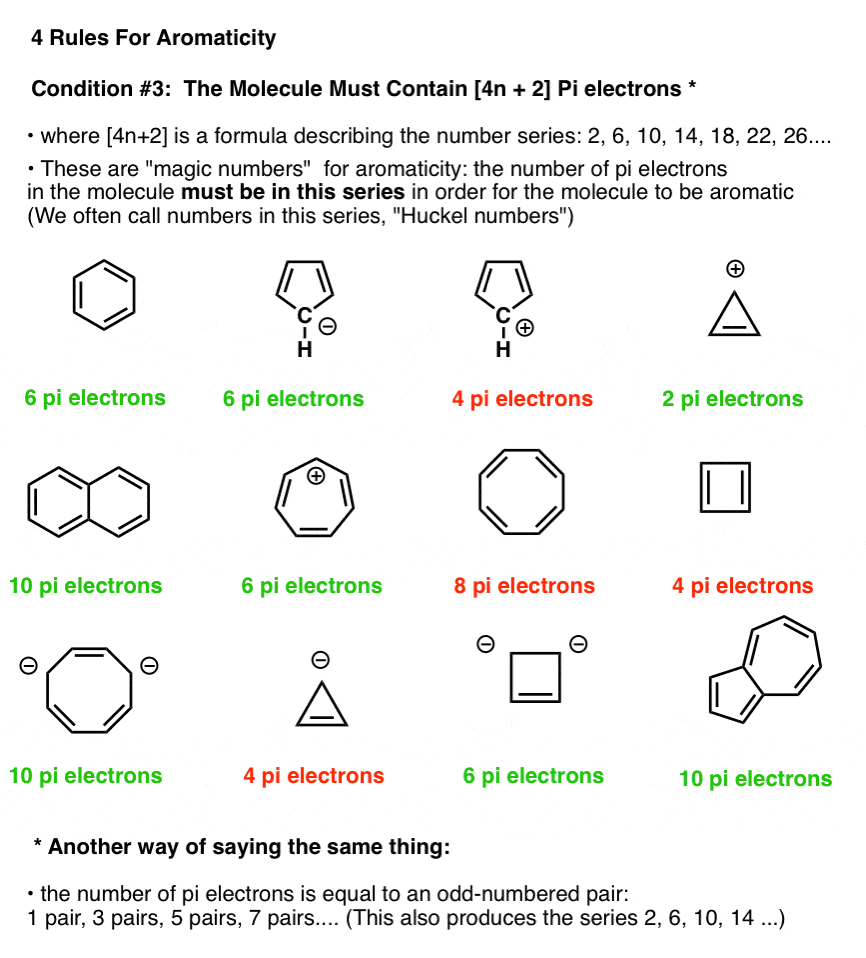
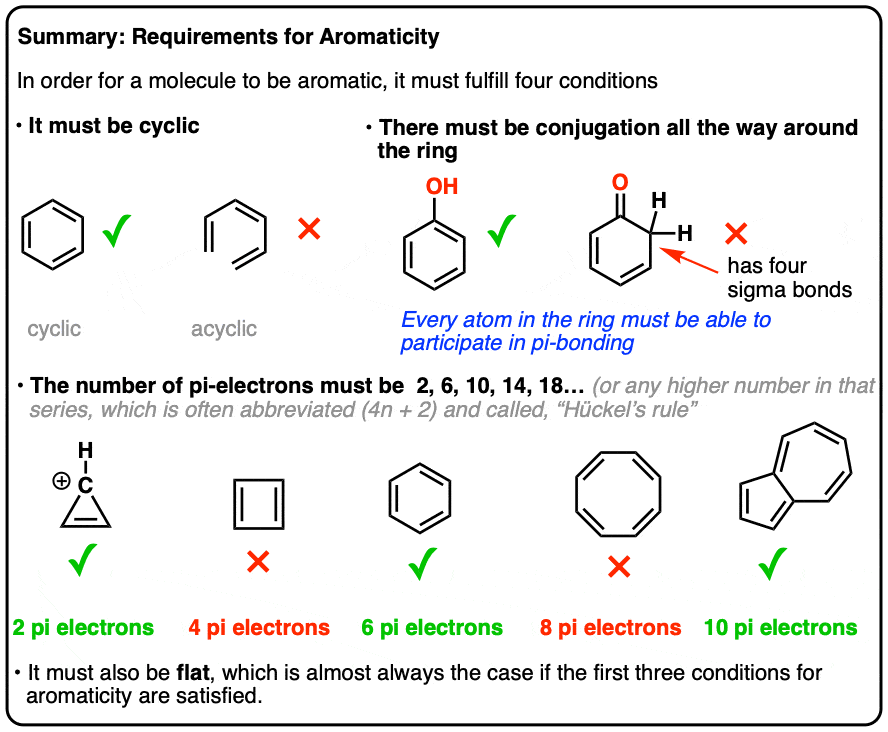
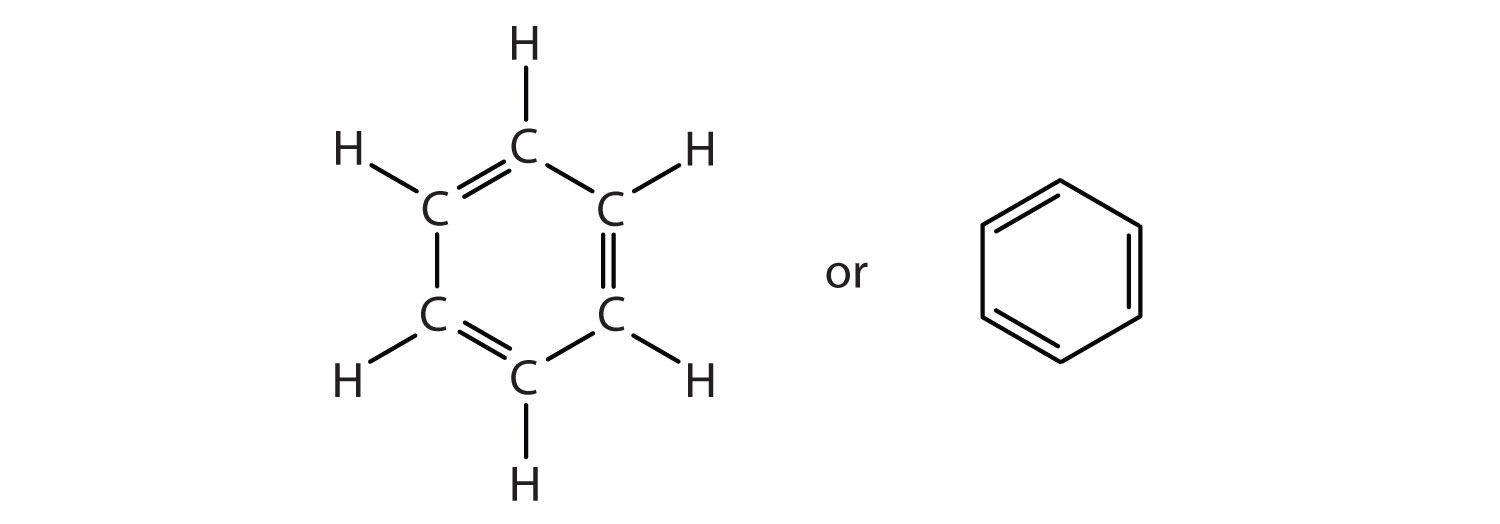
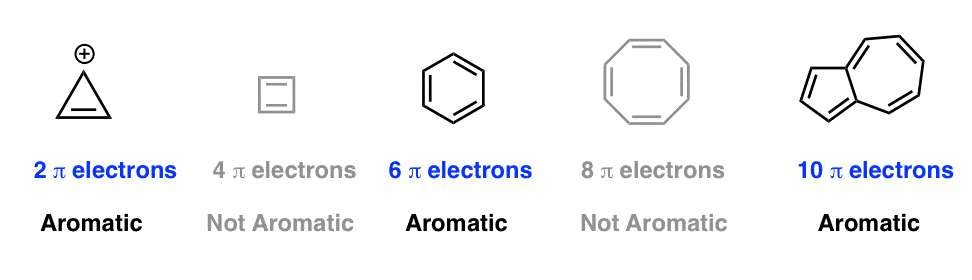
How many electrons does each of the four nitrogen atoms in purine contribute to the aromatic \pi system? | Homework.Study.com

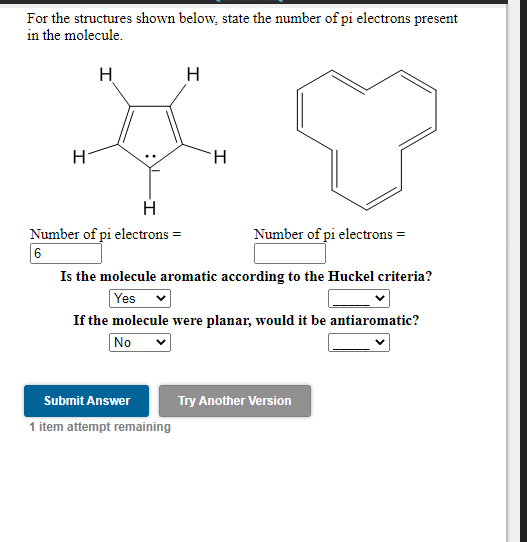
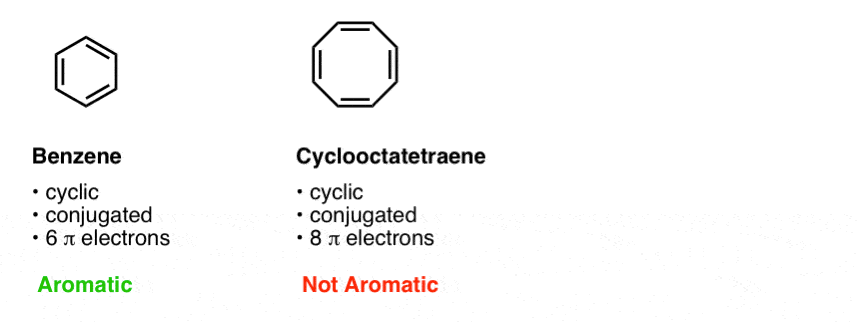
How to identify the number of pi electrons in a conjugated system to calculate the HOMO-LUMO gap with the particle in the box approach? - ECHEMI